Mesalamine tablets are listed in quantities of 10 sachets. What is that quantity in U.S. terms?

Depending on where you are in the world, mesalamine may be available in tablets, capsules, and/or powder sachets. In the U.S., most medications are packaged by the manufacturer in containers with a specific quantity of loose tablets or capsules. It is current practice for the pharmacist to generally dispense prescription medication as loose tablets or capsules that have been transferred from the manufacturer's bottle into a vial by the pharmacy at the time of dispensing.
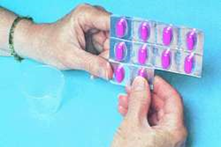
See photos below:
Mesalamine is the generic name and active ingredient in multiple medications in the U.S. and abroad, e.g., Rowasa Enema as well as Pentasa capsules and Asacol HD tablets. Pentasa tablets contain 500 mg or 1 g mesalamine as the active ingredient. Pentasa sachets contain 1 g, 2 g, or 4 g of mesalamine as the active ingredient.
You can compare prices for Mesalamine on PharmacyChecker.com. Currently, prices for Mesalamine 500 mg are as low as $0.48/pill.
However, medications lawfully manufactured in other countries generally have different labeling and packaging than medications approved by the FDA for distribution in the U.S.
Outside the United States, most drugs are packaged by the manufacturer in containers with tablets or capsules enclosed in individual blisters, often in strips of 10 and on flat cards that take up little space. These are usually called blister packs. In most cases, medication ordered from verified international online pharmacies is dispensed in the manufacturer's original sealed package of 100 tablets / capsules in blister pack packaging.
The approved-pharmacies listing Mesalamine tablets on PharmacyChecker.com are required to follow PharmacyChecker Verification Program standards and guidance policies, which permit pharmacies to market, sell, process and /or dispense a maximum 3-months' supply of non-controlled medication pursuant to a valid prescription authorizing a 3-months' supply quantity.